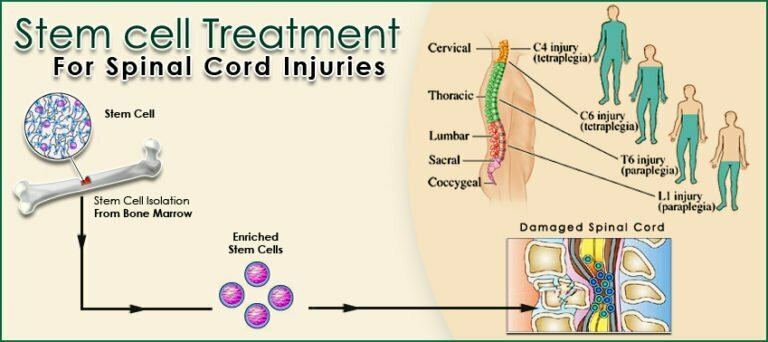
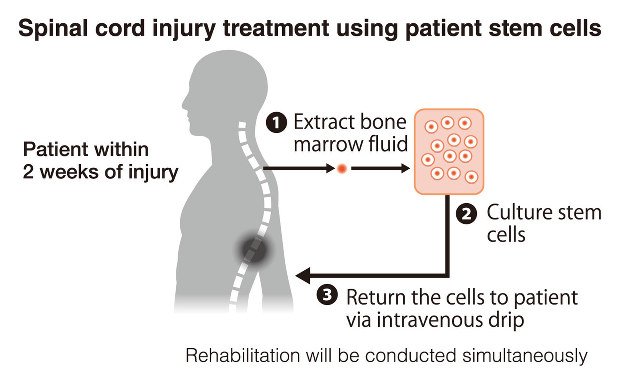
Spinal cord injury (SCI) is one of the most devastating neurological conditions: damage to the spinal cord often leads to permanent loss of motor function, sensation, bowel/bladder control and a major drop in quality of life. Conventional treatments (surgery, decompression, steroids, rehabilitation) help stabilise the injury and maximise residual function—but they do not reliably regenerate damaged spinal neural tissue. PubMed
In recent years, the concept of using stem cells to repair or regenerate injured spinal cords has gathered enormous interest. But what does the evidence say? Can stem-cell therapy truly help in SCI, or is most of it still experimental hype? Let’s explore the current science.
What stem cells bring to the table
Stem cells offer a few key mechanisms for SCI repair:
- They may replace lost or damaged neural cells (neurons, glia) or support the survival of remaining ones. PubMed+1
- They can secrete neurotrophic (growth) factors, modulate inflammation, and create a more favourable environment for regeneration. BioMed Central+1
- They may integrate into scaffolds or support biomaterials and help reconnect neural circuits, though this is much further behind in development. PubMed+1
Various types of stem cells have been studied: mesenchymal stem cells (MSCs) from bone-marrow or umbilical cord, neural progenitor/stem cells, induced-pluripotent stem cells, embryonic stem cells, and newer cell types (e.g., muse cells). PubMed+1
What the human clinical evidence shows
Several systematic reviews and meta-analyses have summarised the data from human trials:
- A meta-analysis of 62 clinical trials involving 2,439 patients found that about 48.9% of patients improved by at least one grade on the ASIA Impairment Scale (AIS) after stem-cell therapy; urinary function improved in ~42%; gastrointestinal/bladder improvements ~52%. PubMed+1
- Another analysis of MSC therapy in 670 patients found statistically significant improvements in AIS grade, sensory scores, bladder function and SSEP (somatosensory evoked potentials) — but not motor score or activities of daily living. PubMed
- A systematic review of MSC therapy in traumatic SCI reported that of 22 studies (21 trials + 1 case series) improvements in AIS grades and sensory scores were consistently seen; motor improvements less so. PubMed
- A recent early-phase clinical trial of intravenous allogeneic “Muse” cells in cervical SCI (10 participants) found good tolerability and some improvements in motor score and quality of life—but this was a non-randomised, non‐blind, single-arm study, so efficacy remains uncertain. BioMed Central
So what is the “truth” behind it?
Positive signs:
- There is human evidence showing that stem cell therapy can lead to measurable neurological improvement (sensory, bladder, AIS grade) in a substantial subset of patients.
- Safety so far appears acceptable in many trials: no major tumour-formation or catastrophic adverse events reported in most reviews. PubMed+1
- The field continues to evolve – new cell types, improved delivery methods (scaffolds, biomaterials), combinations of therapy are underway.
Important caveats / limitations:
- Many trials are small, uncontrolled (single-arm), non‐blinded; thus risk of bias is high. BioMed Central+1
- Heterogeneity is large: different cell types, doses, delivery routes, timings after injury. There is no standard protocol yet. PubMed
- Motor recovery (regaining strength, walking) remains much harder to achieve; most benefit has been in sensory, bladder/bowel, or AIS grade change rather than dramatic restoration of motor power. PubMed+1
- Long-term data are limited: e.g., the risk of delayed adverse events (tumours, graft overgrowth) is still not fully ruled out because follow-up often is modest. PubMed
- Cost, accessibility, ethical/regulatory issues: many “stem-cell clinics” operate outside rigorous trial frameworks and may over-promise.
What this means in practice
For clinicians and patients alike: stem cell therapy for SCI is promising, but it is not yet a proven regenerative cure. At present it is best considered experimental. If one is considering it, the following are sensible points:
- Prefer enrolment in a well-designed clinical trial rather than unregulated “commercial” stem-cell treatments.
- Timing may matter: many studies suggest that sub-acute phase (not too chronic) may give better results. PubMed
- Understand realistic expectations: sensory or bladder improvement is more likely; full motor recovery remains rare.
- Rehabilitation remains essential: stem cells will not replace the need for rigorous physical therapy and supportive care.
- Follow-up, monitoring, and long-term safety matter.
- Policymakers and clinicians should work towards standardising cell type, dose, delivery, timing, and outcome measures to improve comparability and evidence quality.
Conclusion
In short: yes, stem cell therapy in spinal cord injury has moved from “wild hope” to credible scientific investigation. The evidence indicates moderate improvement in many cases—especially sensory and autonomic functions—but full motor regeneration remains elusive. It’s a field full of potential, but also full of challenges. Until large, well-designed, controlled trials become available, we must remain cautiously optimistic rather than over-enthusiastic.